 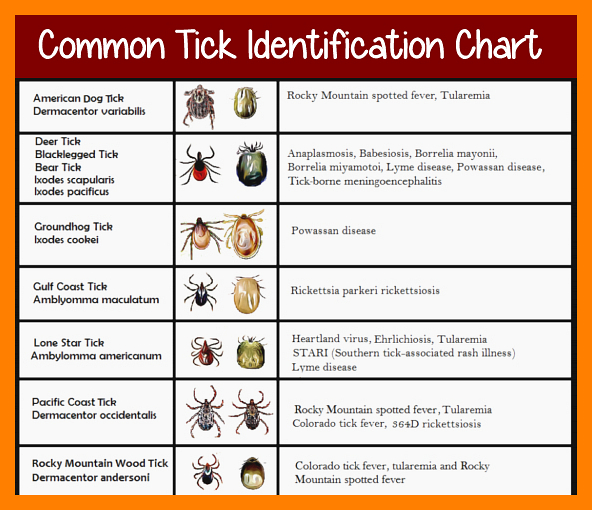 
The result is that passive surveillance programs produce reliable data about whether vector tick species are present in an area, the proportion of ticks infected with a tick-borne pathogen (limited in geographic granularity in regions from which few ticks are submitted), and the progress of establishment as a nonendemic tick species spreads into a territory not colonized previously. Passive surveillance programs relying on self-reporting by tick-bite victims or their physicians are comparatively easily and cheaply maintained, producing datasets of far greater scope but substantially less precision than active surveillance programs. These programs, however, are expensive in resources and manpower, which typically results in surveillance efforts that are quite limited in scope and duration. 2016).Īctive surveillance programs emphasizing systematic collection and pathogen testing of vector ticks provide great precision in evaluating the spatial and temporal distribution of ticks and patterns of pathogen distribution. scapularis in the midwestern and northeastern United States, and the concomitant expansion of Lyme disease, emphasize the increasing importance of this disease to public health ( Lingren et al. Furthermore, the expanding distribution of I. Although very few fatalities result directly from Lyme disease infection, the number of people infected and morbidity inflicted underscores the public health importance of this illness. Of these, Lyme disease is by far the most significant, with about 30,000 cases reported to the CDC per year ( ) and an estimated 10 times as many cases diagnosed ( Hinckley et al. It transmits a variety of disease-causing organisms, including those responsible for Lyme disease, human granulocytic anaplasmosis, babesiosis, and Powassan virus. The blacklegged tick, Ixodes scapularis Say, is currently the most important vector of disease to humans in the continental United States. Tick, Rickettsia buchneri, Anaplasma phagocytophilum, Lyme disease The results of these surveillance programs indicate an increasing risk of disease transmission by I. phagocytophilum, and 67.3% for Rickettsia spp. (likely representing Rickettsia buchneri). burgdorferi, Anaplasma phagocytophilum, and spotted fever group Rickettsia spp. Ixodes scapularis nymphs collected at these sites were tested for the presence of B. Active surveillance was performed at selected sites from 2007–2009. burgdorferi, the highest proportion of any year. burgdorferi increased over time between 19. The proportion of vector ticks infected by B. scapularis per year over the surveillance period, indicating expansion of this tick species across the state. An average of 2.6 of Iowa’s 99 counties submitted first reports of I. Submitted ticks were identified to species and life stage, and Ixodes scapularis Say nymphs and adults were tested for the presence of Borrelia burgdorferi. andersoni and comments on its seasonal occurrence.A passive surveillance program monitored ticks submitted by the public in Iowa from 1990–2013. Based upon the database collection records, we also present a summary of recorded hosts for D. 
The data will be useful for identifying regions at increased risk of acquiring anaplasmosis in the United States. andersoni populations in the United States currently extend from the western portions of Nebraska and the Dakotas westward to the Cascade Mountains and from the northern counties of Arizona and New Mexico northward to the Canadian border. andersoni and 87 counties with reported occurrences in 14 states with the majority of established populations reported from Montana, Idaho, and Oregon. We found 180 counties with established populations of D. andersoni were recorded from 267 counties in 14 states and were distinguished as either established or reported. The records spanned the period from 1903 through 2001 with the majority between 1921 to 1940. We reviewed the geographic records in each database and postings from MEDLINE and AGRICOLA to produce a national county-level distribution map based on a total of 5,898 records. andersoni from two large national databases. We developed a centralized geographic database that incorporates collection records for D. The risk of transmission of anaplasmosis to cattle has been largely based on the distribution of D. In the United States, Dermacentor andersoni Stiles and Dermacentor variabilis (Say) are the principal vectors. There are approximately 20 tick species worldwide that are implicated as vectors of this pathogen. Anaplasma marginale Theiler is a tick-borne pathogen that causes anaplasmosis in cattle. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
